Peer Reviewed
A Healthy Male With Recalcitrant Herpes Zoster
Case Report Insights: For an in-depth look at the Photoclinic case report below and an interview with the authors, visit https://www.consultant360.com/videos/recurring-herpes-zoster-healthy-patient
A 62-year-old healthy man presented to his primary care provider with a 2-day history of painful vesicular rashes on his right trunk.
History. The patient noticed a burning sensation in his right trunk for 6 days followed by painful vesicular rashes in the same area for the next 2 days. He denied any other symptoms. He had no significant medical history other than chickenpox in childhood. He did not take any medication. His physical examination was unremarkable except for a large area of baseline erythema with overlying tense vesicles extending across the right upper back and right chest in a T5 to T7 dermatomal distribution (Figure 1).
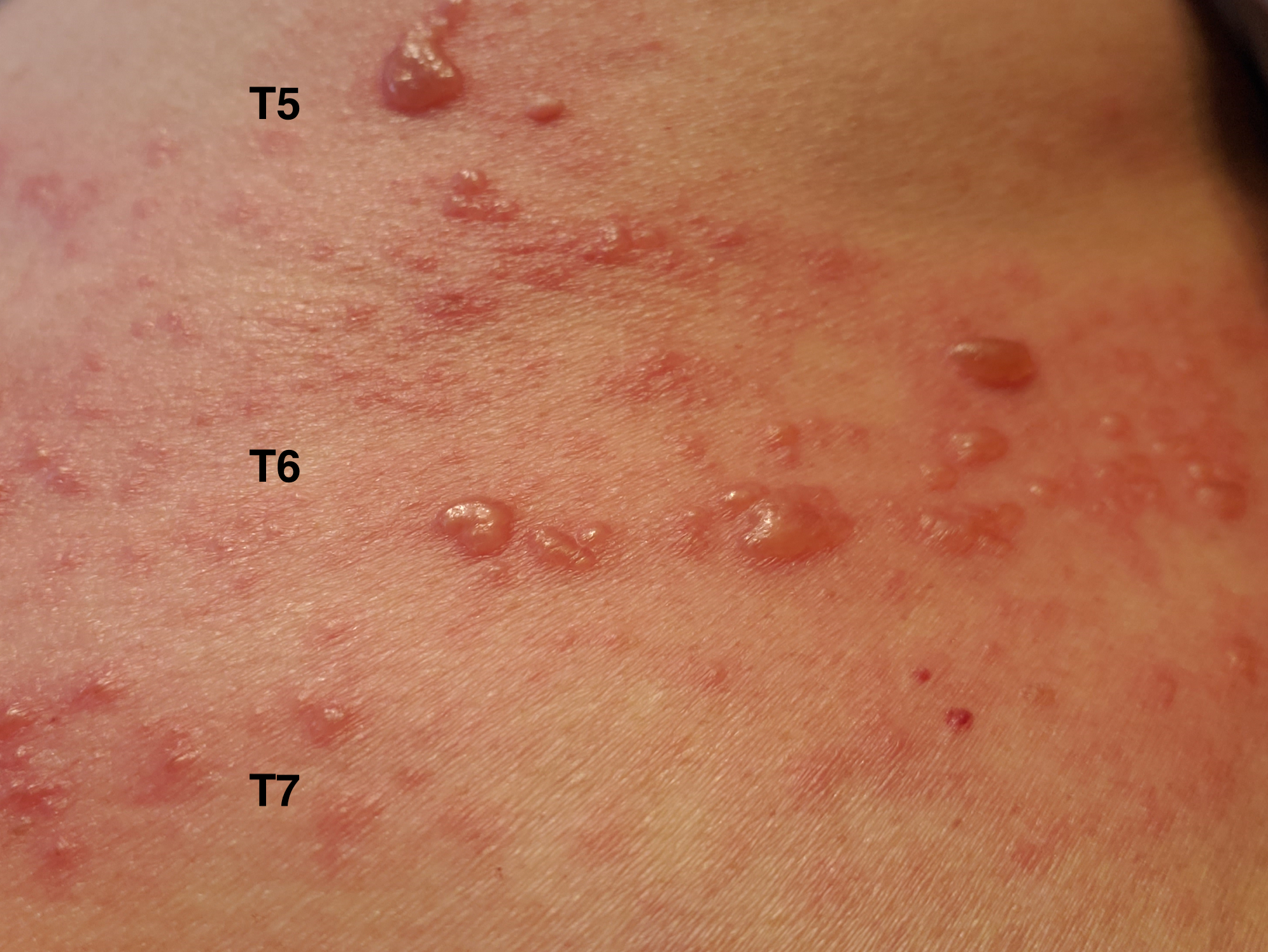
Figure 1. Unilateral vesicular rash in the T5 to T7 dermatomes on the right upper back and right chest.
Diagnostic testing. Based on the history and physical examination findings, the patient was diagnosed with herpes zoster (HZ).
Differential diagnoses. Herpes simplex virus (HSV) infection and contact dermatitis were considered in the differential diagnosis due to the erythematous vesicular appearance of the rash. Although HSV infection also causes a painful unilateral vesicular rash, HSV typically affects the trigeminal or sacral ganglia resulting in oral/labial or genital lesions. Other cutaneous manifestations of HSV infection include herpetic whitlow on fingers, eczema herpeticum for patients with a history of eczema, and herpes gladiatorum on the face, neck, and arms of wrestlers. The location of the rash on the trunk and no prior history of eczema in this patient made HSV infection unlikely. A lack of contact with foreign substances and the fact that the lesions were painful and not pruritic made contact dermatitis also unlikely.
Treatment and management. The patient was treated with valacyclovir 1000 mg three times daily for 7 days. The pain improved and the rash fully crusted. Two days after finishing valacyclovir, the burning sensation recurred followed by the appearance of a few new vesicles in the same area (Figure 2). He was treated with another course of valacyclovir 1000 mg three times daily for 7 days with a resolution of symptoms. Two weeks after completion of the second course of valacyclovir, the burning sensation recurred on the right chest slightly above the previous rash area in the T3 to T4 dermatomes (Figure 3). He was treated with a third course of valacyclovir 1000 mg three times daily for 7 days, followed by 500 mg valacyclovir daily for 3 to 4 weeks to prevent recurrence. The symptoms resolved at the end of the 7-day course of treatment.
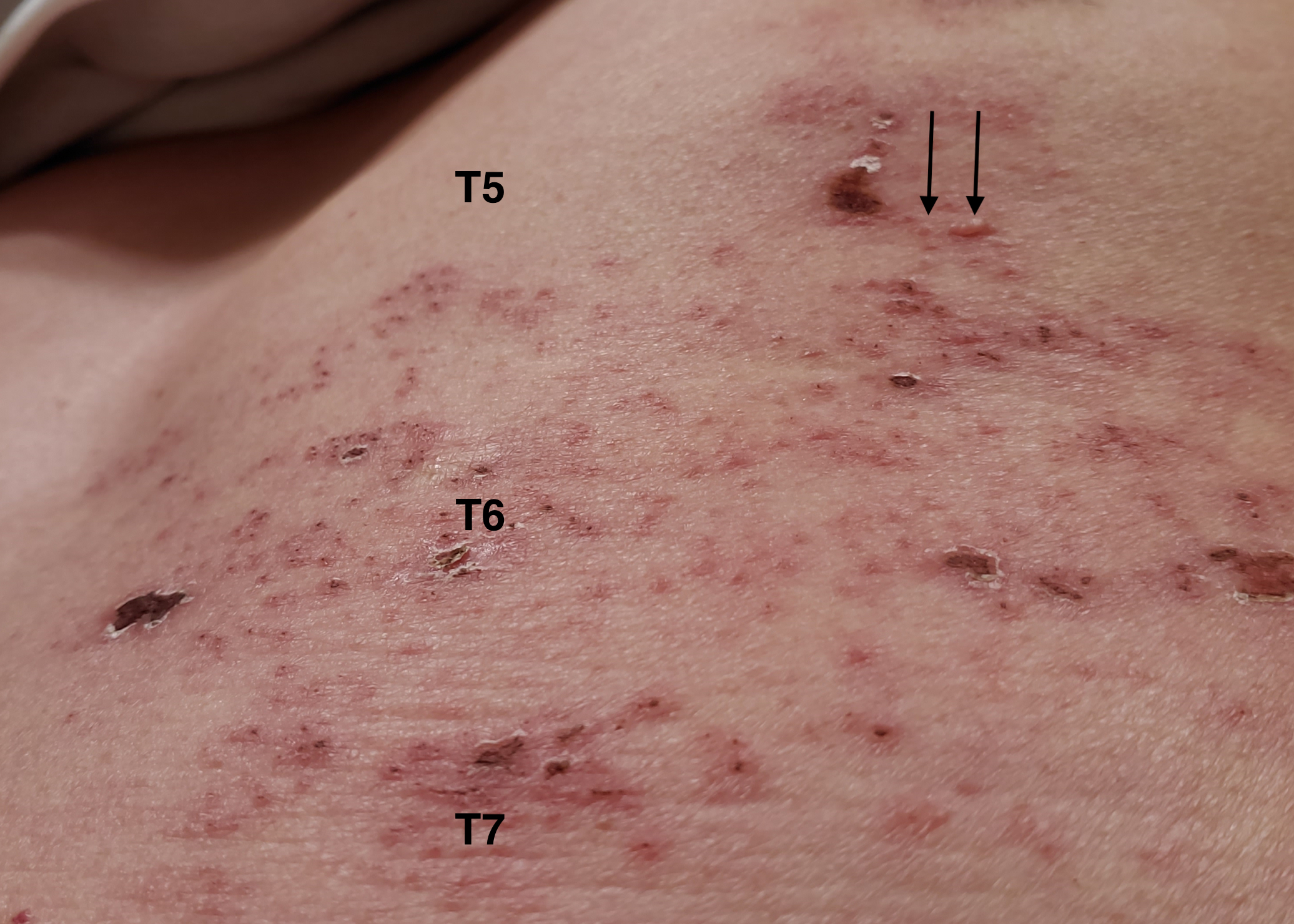
Figure 2. New vesicular rash 2 days after completion of the first 7-day course of valacyclovir (arrow heads).
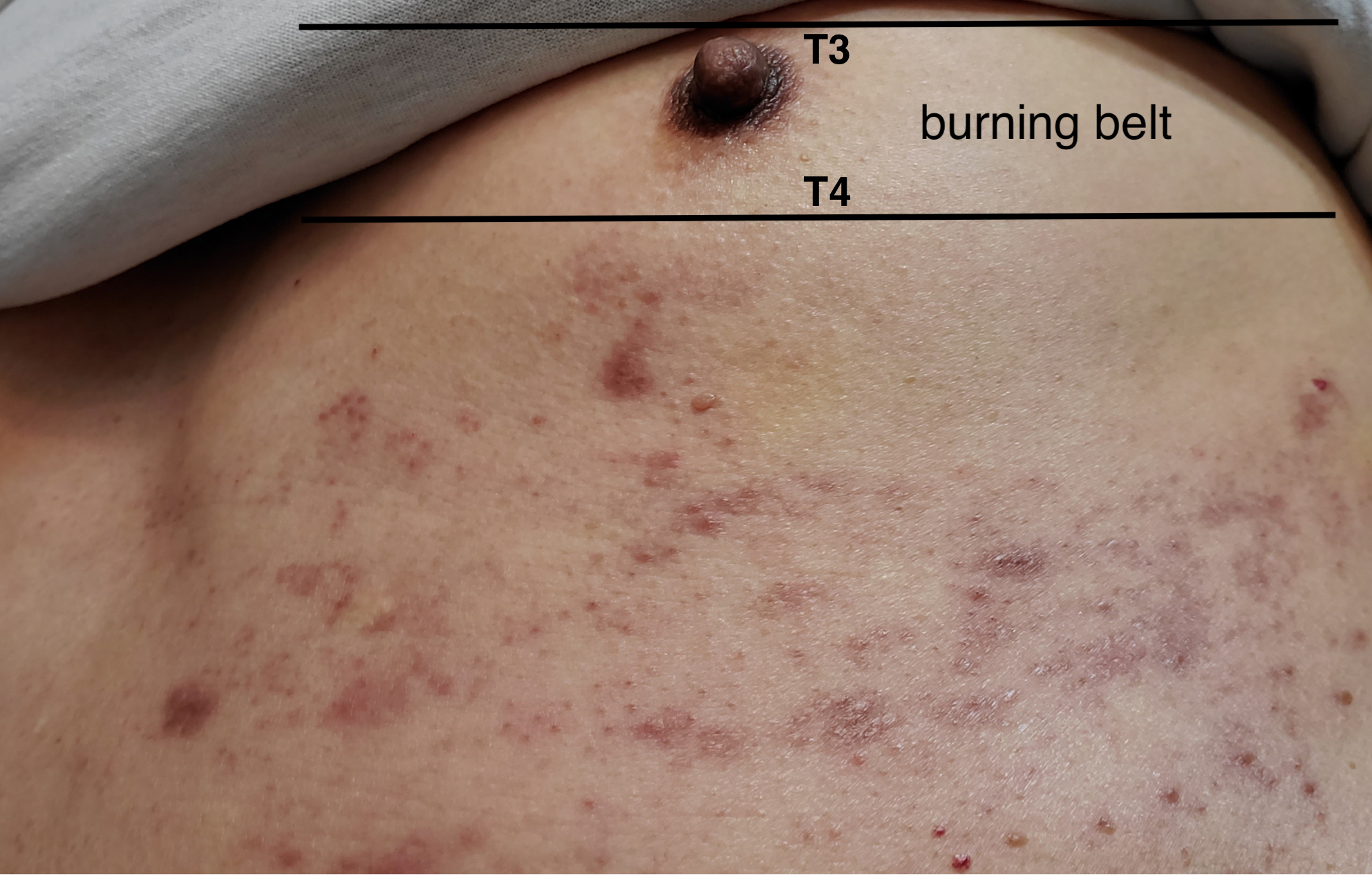
Figure 3. New burning sensation belt in the T3 to T4 dermatomes 2 weeks after completion of the second 7-day course of valacyclovir.
However, two weeks after the resolution of symptoms, the burning sensation recurred in the same areas as the previous symptoms while the patient was taking valacyclovir 500 mg daily. Due to the recurrence of symptoms despite three courses of standard antiviral treatment, evaluation for immunodeficiency was recommended but the patient declined. He was referred to an infectious disease specialist who suspected undertreated HZ and prescribed valacyclovir 1000 mg three times daily for 2 weeks.
Outcome and follow-up. The 2-week course of valacyclovir resolved his symptoms. He elected to receive a varicella zoster recombinant, adjuvanted vaccine 4 months after the last HZ episode. The patient did not have recurrent burning or rash at his routine follow-up visit 12 months after the first episode of HZ.
Discussion. HZ, also known as shingles, is a common condition encountered by primary care providers. HZ is caused by reactivation of latent varicella zoster virus (VZV) that gains access to sensory ganglia during initial infection.1 It is characterized by painful, unilateral vesicular eruptions in one to two contiguous dermatomes, most commonly the thoracic and lumbar dermatomes.1 In immunocompetent hosts, the lesions crust by 7 to 10 days and heal within 2 to 4 weeks.1 Neuropathic pain such as aching, burning, and lancinating often proceeds rash onset over the involved dermatomes and may be associated with systemic symptoms of fever, fatigue, headache, malaise, and photophobia.1 The most common complication is postherpetic neuralgia (PHN), which may last for months to years.1
In immunocompetent individuals, the diagnosis of uncomplicated HZ is usually based solely on the clinical presentation of unilateral painful vesicular eruptions within well-defined dermatomes.1,2 Antiviral therapy is recommended for patients who present within 72 hours of clinical symptoms or after 72 hours if new lesions arise at the time of presentation.1 Conventional regimens include valacyclovir 1000 mg three times daily for 7 days, famciclovir 500 mg three times daily for 7 days, or acyclovir 800 mg five times daily for 7 to 10 days.1,2,3,4 While antiviral therapy is effective in accelerating the healing of cutaneous lesions, decreasing the duration and severity of acute neuritis, and reducing acute pain,1,2,3,4 it does not decrease the risk of PHN.5
Two vaccines are licensed to prevent both HZ and PHN: a live zoster vaccine and a recombinant, adjuvanted zoster vaccine.6 The live-attenuated vaccine is indicated for immunocompetent adults aged 60 years and older.6 The overall efficacy is 51.3% against HZ and 66.5% against PHN.6 The protection duration against HZ is up to 8 years.6 Due to its low efficacy and diminished effectiveness over time, the live zoster vaccine was discontinued in 2020 in the United States. The recombinant, adjuvanted vaccine is indicated for immunocompetent adults aged 50 years and older, as well as immunocompromised adults aged 19 years and older. 6,7 The overall efficacy is 97.2% against HZ and 91.2% against PHN.6,7 The protection duration against HZ is more than 10 years.6 The recombinant, adjuvanted vaccine is administered in two doses given 2 to 6 months apart.8 There is no specific amount of time to wait before administering the recombinant vaccine to patients who have had HZ. However, patients should not receive the recombinant vaccine when they are experiencing an acute episode of HZ.8
This case highlights an unusual presentation of acute zoster symptoms in an otherwise healthy man that recurred three times, each time within 2 weeks after a standard 7-day course of valacyclovir treatment. The reasons that a patient with HZ fails to respond to conventional antiviral treatment may include acyclovir-resistant HZ virus (HZV), an underlying immunodeficiency, chronic zoster, or undertreatment of the condition. Although acyclovir-resistant HZ has been identified in high-risk immunocompromised patients on long-term acyclovir therapy,9 it is rare in immunocompetent populations.9 The fact that the lesions resolved after each treatment and recured within 2 weeks of treatment makes acyclovir-resistant HZ virus less likely.
Patients with underlying immunodeficiency may develop new lesions more than a week after presentation.1 Our patient was otherwise healthy and at low risk for immunodeficiency. Nevertheless, he was offered evaluation for immunodeficiency but declined. Typically, cellular immunity deficiency increases the risk of recurrent viral infections, including HZ.10 VZV cell-mediated immune response involves T lymphocytes, including CD4+ and CD8+ effector and memory T cells, as well as natural killer cells.10 The initial immunodeficiency evaluation should include complete blood count with differential and HIV testing.11,12,13 If absolute lymphocytes count is low, flow cytometry can further identify the subsets of T cells (CD3, CD4, and CD8) and natural killer cells (CD16 and CD56).11,12,13 A referral to an allergist/immunologist for further evaluation can be done if needed.
Currently, there is no literature that reports on chronic cutaneous zoster, although chronic herpes zoster ophthalmicus is not uncommon.14,15 Risk factors for herpes zoster ophthalmicus include conjunctivitis, epithelial keratitis, and stromal keratitis.14 The duration of antiviral treatment does not affect the course of this disease.14,15
The fact that longer course of valacyclovir treatment was successful for lasting resolution of symptoms suggests that our patient was undertreated initially with the conventional 7-day treatment. Most patients with HZ have a rash over 1 to 2 dermatomes.1 Interestingly, at least four contiguous dermatomes and large areas from back to chest were involved in this case. It is possible this may denote a larger burden of disease. In such cases, physician may consider longer treatment strategies. However, further research is needed for the optimal treatment duration for acute HZ involving large areas of skin and multiple dermatomes.
- Dworkin RH, Johnson RW, Breuer J, et al. Recommendations for the management of herpes zoster. Clin Infect Dis. 2007;44 Suppl 1:S1-26. doi:10.1086/510206
- Patil A, Goldust M, Wollina U. Herpes zoster: a review of clinical manifestations and management. Viruses. 2022;14(2):192. doi:10.3390/v14020192
- Pott Junior H, de Oliveira MFB, Gambero S, Amazonas RB. Randomized clinical trial of famciclovir or acyclovir for the treatment of herpes zoster in adults. Int J Infect Dis. 2018;72:11-15. doi:10.1016/j.ijid.2018.04.4324
- Tyring SK, Beutner KR, Tucker BA, Anderson WC, Crooks RJ. Antiviral therapy for herpes zoster: randomized, controlled clinical trial of valacyclovir and famciclovir therapy in immunocompetent patients 50 years and older. Arch Fam Med. 2000;9:863-869. doi:10.1001/archfami.9.9.863
- Chen N, Li Q, Yang J, Zhou M, Zhou D, He L. Antiviral treatment for preventing postherpetic neuralgia. Cochrane Database Syst Rev. 2014;(2):CD006866. doi:10.1002/14651858.CD006866.pub3
- Harbecke R, Cohen JI, Oxman MN. Herpes zoster vaccines. J Infect Dis. 2021;224(12 Suppl 2):S429-S442. doi:10.1093/infdis/jiab387
- About the vaccine. Centers for Disease Control and Prevention. Updated January 22, 2022. Accessed April 25, 2024. www.cdc.gov/vaccines/vpd/shingles/hcp/shingrix/about-vaccine.html
- Shingrix recommendations. Centers for Disease Control and Prevention. Updated January 24, 2022. Accessed April 25, 2024. www.cdc.gov/vaccines/vpd/shingles/hcp/shingrix/recommendations.html
- Piret J, Boivin G. Antiviral resistance in herpes simplex virus and varicella-zoster virus infections: diagnosis and management. Curr Opin Infect Dis. 2016;29(6):654-662. doi:10.1097/QCO.0000000000000288
- Laing KJ, Ouwendijk WJD, Koelle DM, Verjans GMGM. Immunobiology of varicella-zoster virus infection. J Infect Dis. 2018;218(suppl 2):S68-S74. doi:10.1093/infdis/jiy403
- Azar AE, Ballas ZK. Evaluation of the adult with suspected immunodeficiency. Am J Med. 2007;120(9):764-768. doi:10.1016/j.amjmed.2006.12.013
- Fleisher TA. Evaluation of suspected immunodeficiency. Adv Exp Med Biol. 2007;601:291-300. doi:10.1007/978-0-387-72005-0_31
- Reust CE. Evaluation of primary immunodeficiency disease in children. Am Fam Physician. 2013;87(11):773-778
- Lee SM, Han J, Yang CM, et al. Chronic and recurrent herpes zoster ophthalmicus. Medicina. 2021;57(10): 999. doi:10.339/medicina57100999
- Lo DM, Jeng BH, Gillespie C, Wu M, Cohen EJ. Current practice patterns and opinions on the management of recent-onset or chronic herpes zoster ophthalmicus of zoster eye disease study investigators. Cornea. 2019;38(1):13-17. doi:10.1097/ICO.0000000000001732
AFFILIATIONS:
1Professor, Department of Family & Community Medicine, Pennsylvania State University College of Medicine, State College, PA
2Medical Student, University of Pittsburgh School of Medicine, Pittsburgh, PA
3Associate Professor, Department of Dermatology, Pennsylvania State University College of Medicine, State College, PA
CITATION:
Qiu J, Lin A, Ferguson S. A healthy male with recalcitrant herpes zoster. Consultant. 2024;64(5):e2. doi:10.25270/con.2024.05.000001
Received December 8, 2023. Accepted February 23, 2024. Published online May 9, 2024.
DISCLOSURES:
The authors report no relevant financial relationships.
ACKNOWLEDGEMENTS:
None.
CORRESPONDENCE:
Juan Qiu, MD, PhD, Pennsylvania State University College of Medicine, Penn State Health Medical Group, 32 Colonnade Way, State College, PA 16803 (jqiu@pennstatehealth.psu.edu)


